|
While the use of intrathecal opioids by continuous infusion has been well studied ( 10), few studies have examined the frequency of clinically significant secondary adrenal insufficiency (SAI) arising from oral or transdermal opioids. This observation led to the notion that ACTH was under the influence of inhibitory central opioid tone, and the extent of central opioid tone could be quantified by the magnitude of the ACTH rise ( 9). In contrast, high-dose naloxone, an opioid receptor antagonist, results in an increase in ACTH and cortisol ( 8). With respect to the hypothalamic–pituitary–adrenal (HPA) axis, early studies demonstrated that acute IV morphine and other opioids lowered adrenocorticotropic hormone (ACTH) and cortisol ( 7). Less is known about the chronic effects of prolonged oral and transdermal opioid analgesic use on hypothalamic–pituitary function. This effect is thought to be mediated via inhibition of the relevant hypothalamic releasing factors by opioid receptors which are widely distributed throughout the central nervous system ( 4, 5, 6).  
It has been known for several decades that acute administration of opioids intravenously (IV) to humans alters several pituitary hormones ( 3). Adverse effects are common, including drowsiness, gastrointestinal disturbance and an increase in mortality ( 2). 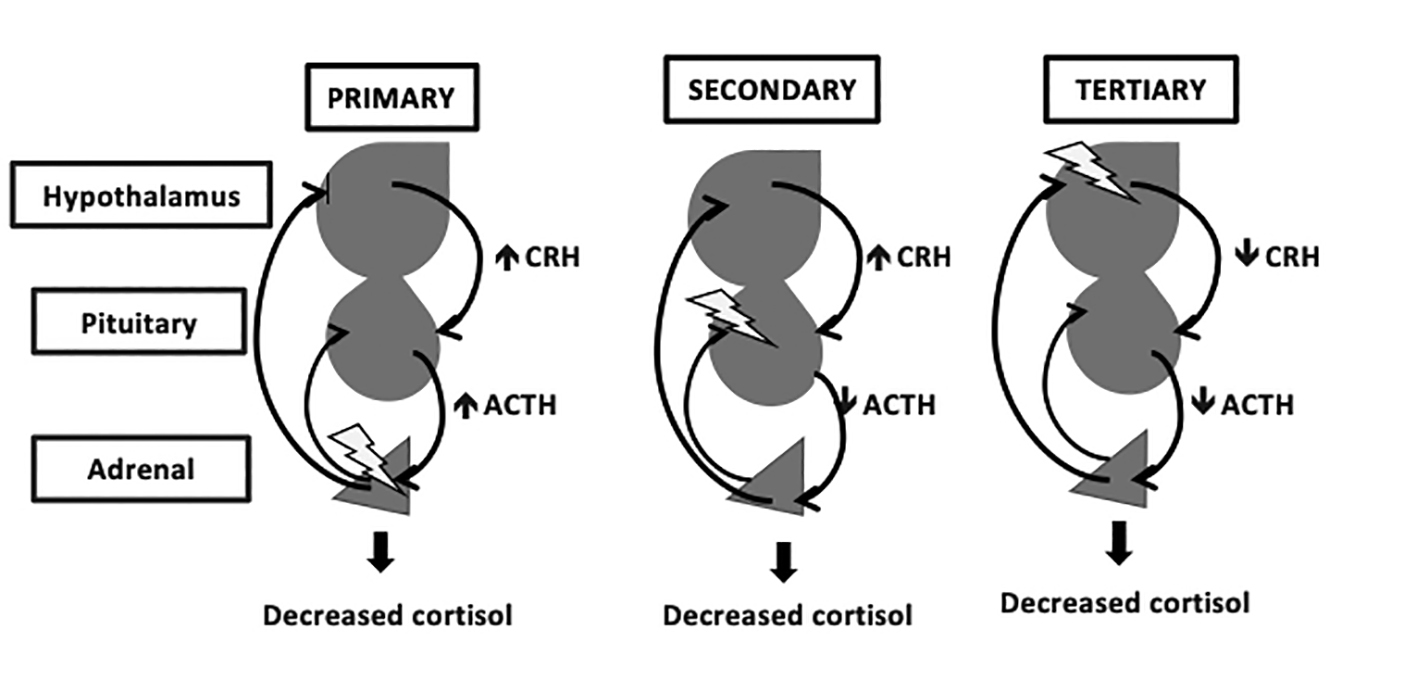
The use of opioid analgesics, particularly oxycodone, for non-cancer pain is increasing in many countries including Australia ( 1).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
